Z6_EL9QHKG10GPM20IJGE61AK10O0
Z7_P86CH0O0I0RF10IB06HRVQ0087
 S2K Commerce - Products Dropdown
S2K Commerce - Products Dropdown
Z7_60E2I18209KBD06G17A9D51006
 Web Content Viewer
Web Content Viewer
Z7_EL9QHKG10GPM20IJGE61AK10S5
 S2K Commerce - Order Entry
S2K Commerce - Order Entry
You must be logged in to order the below items. Log in to order
Baxter Parenteral Products: CLINIMIX E 4.25/25- Sulfite- Free (4.25% Amino Acid in 25% Dextrose) Injection without Electrolytes in CLARITY Dual Chamber Container. Fill (mL): 1000, Container Size (mL): 2000, Units per case: 6.
Item#:
114956
Manufacturer Item:
2B7729
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
51191603
Baxter Parenteral Products: CLINIMIX E 4.25/10- Sulfite- Free (4.25% Amino Acid in 10% Dextrose) Injection without Electrolytes in CLARITY Dual Chamber Container. Fill (mL): 1000, Container Size (mL): 2000, Units per case: 6.
Item#:
114957
Manufacturer Item:
2B7727
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
51191603
Item#:
116930
Manufacturer Item:
1C8740
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42221616
Item#:
146360
Manufacturer Item:
70402609V1
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42201862
Adapter converts standard luer lock syringes to a catheter tip. Safely transport drugs in a securely capped standard luer lock syringe, then convert the syringe to a catheter tip for administration. Specifications: PP (Polypropylene). Packaging:50/Case.
Item#:
147852
Manufacturer Item:
H93813601
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42142615
Item#:
154646
Manufacturer Item:
2C4005
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42293509
Item#:
155790
Manufacturer Item:
2C8891
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42221902
Item#:
155842
Manufacturer Item:
2C6226
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42221616
EVA (ethyl vinyl acetate) for total parenteral nutrition (TPN) production with large-bore threaded fill port connector. Safe for 3-in-1 compounding. EVA is environmentally friendly and stable for medical solutions. Specifications: Bag: EVA (ethyl vinyl acetate). Fill Port: ABS (acrylonitrile butadienestyrene). Additive Port: Latex-free injection port. Clamp: Polypropylene. 2000ML. Sterile. Packaging:50/Case.
Item#:
156342
Manufacturer Item:
H938740
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42221701
Item#:
156890
Manufacturer Item:
2C4014
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42222202
Item#:
156964
Manufacturer Item:
2B2544X
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
51191704
Item#:
158052
Manufacturer Item:
2C8402
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42221609
EVA (ethyl vinyl acetate) for total parenteral nutrition (TPN) production with large-bore threaded fill port connector. Safe for 3-in-1 compounding. EVA is environmentally friendly and stable for medical solutions. Specifications: Bag: EVA (ethyl vinyl acetate). Fill Port: ABS (acrylonitrile butadienestyrene). Additive Port: Latex-free injection port. Clamp: Polypropylene. 500ML. Sterile. Clear. Packaging:50/Case.
Item#:
158085
Manufacturer Item:
H938738
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42221701
Tamper-Evident Tip Cap assures your drugs are delivered where they should be. Caps are an easy-to-use luer lock, latex-free connection. Specifications: Cap: PP (Polypropylene). Sleeve: Red polystyrene. Sterile. Packaging:1000/Case.
Item#:
172093
Manufacturer Item:
H93864110
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42142615
Extension Set with 0.22 micron downstream high pressure extended life filter, polyehtylene lined tubing and male Luer Lock adapter. Sterile package. Filer has passed bacterial and pyrogen retention testing for 96 hours. Fluid path is non-DEHP. 48 units/case.
Item#:
193169
Manufacturer Item:
1C8363
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42221607
Item#:
202031
Manufacturer Item:
2B0962
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
51271629
Empty INTRAVIA Container with Non-DEHP Fluid Path. INTRAVIA Container, 500 mL.UOM: Case/48.
Item#:
202232
Manufacturer Item:
2J8003
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42221701
Item#:
234678
Manufacturer Item:
2B6065
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
50501803
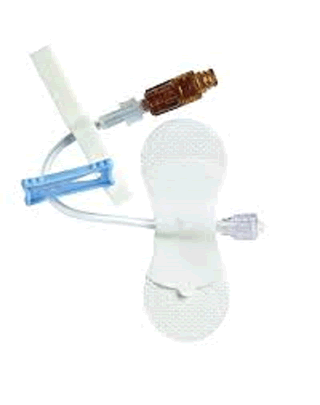
Baxter Catheter Stabilization Device Extension Set Kit. V-LINK Device with VITALSHIELD Protective Coating Non-DEHP Catheter Extension Set Kit, Standard Bore Tubing, 1 luer activated device with silver for IV Access, Medium VITAL HOLD Catheter Stabilization Device, foam adhesive strips. Holds tubing and lines 3.0 mm to 8.0 mm in diameter. Latex Free. UOM: 50/CS.
Item#:
234884
Manufacturer Item:
6N8220
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42221616
Item#:
253459
Manufacturer Item:
STP5604
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42221612
Item#:
271603
Manufacturer Item:
1C8507
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42192604
0.9% Sodium Chloride Irrigation, USP. in Uromatic Plastic Containers. Container Type: Viaflex. Size: 2000mL. mEq/L: Na 154, Cl 154. pH Range: 5.5 (4.5-7.0). mOsmol/L (calc): 308. Units per case: 6.
Item#:
310298
Manufacturer Item:
2B7126
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
51473504
80mg Isotonic Gentamicin Sulfate Injection, 50ml, Viaflex Bag. 24 units/case. NDC: 0338-0509-41
Item#:
310383
Manufacturer Item:
2B0852
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
51281651
Item#:
326217
Manufacturer Item:
7N8375K
Manufacturer:
BAXTER HEALTHCARE CORPORATION
UNSPSC:
42221616
Item#:
352077
UNSPSC:
42222001
PLEASE CONTACT SALES REP. FOR EQUIPMENT QUOTE